Filters
Question type
A) The rate remains the same.
B) The rate decreases by a factor of 2.
C) The rate increases by a factor of 2.
D) The rate increases by a factor of 4.
E) B) and D)
F) A) and B)
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Question 42
Multiple Choice
Which of the following statements about an SN1 reaction mechanism is true?
A) The reaction is fastest with primary alkyl halide.
B) The reaction exhibits a one-step mechanism.
C) The reaction rate increases as the leaving group ability increases.
D) The reaction rate increases as the strength of the nucleophile increases.
E) A) and C)
F) B) and C)
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Question 43
Multiple Choice
For the following reaction, use the identity of the alkyl halide and nucleophile to determine which substitution mechanism occurs. Then determine which solvent affords the faster reaction. 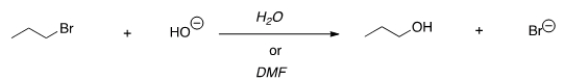
A) SN1, H2O
B) SN1, DMF
C) SN2, H2O
D) SN2, DMF
E) C) and D)
F) A) and C)
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Question 44
Multiple Choice
Which of the following is a polar aprotic solvent? 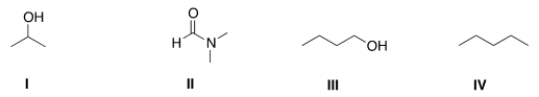
A) I
B) II
C) III
D) IV
E) B) and C)
F) All of the above
F) All of the above
Correct Answer

verified
Correct Answer
verified
Showing 61 - 64 of 64
Related Exams