Correct Answer

verified
 _TB1830_00...
_TB1830_00...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Give the product for the following reaction. 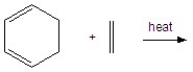
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major product of the following reaction? 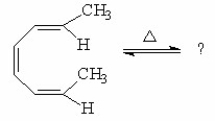
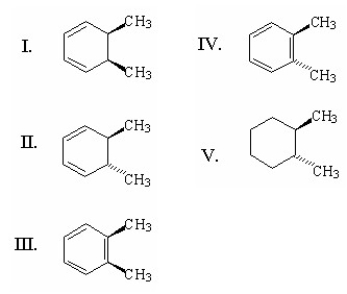
A) I
B) II
C) III
D) IV
E) V
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the allyl radical, which π molecular orbital is singly occupied?
A) the bonding π molecular orbital
B) the nonbonding π molecular orbital
C) the antibonding π molecular orbital
D) none of the above
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Essay
What type of pericyclic process is occurring in the reaction below? Provide two reasons why the equilibrium lies in the direction indicated. 
Correct Answer

verified
A [3,3] sigmatropic rearrangement is occ...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
Under thermal conditions, is an [8+2] cycloaddition suprafacial or antarafacial?
Correct Answer

verified
Correct Answer
verified
Essay
Why does the diene shown below fail to undergo a Diels-Alder reaction with even the most reactive dienophiles? 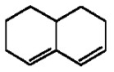
Correct Answer

verified
The diene cannot ach...View Answer
Show Answer
Correct Answer
verified
View Answer
Essay
Give a representation of the bonding π molecular orbital of the allyl cation.
Correct Answer

verified
Correct Answer
verified
Essay
Give a representation of the antibonding π molecular orbital of the allyl anion.
Correct Answer

verified
Correct Answer
verified
Essay
What diene and dienophile would react to give the product below? 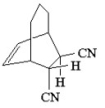
Correct Answer

verified
Correct Answer
verified
Essay
Provide the major organic product of the following Diels-Alder cycloaddition. 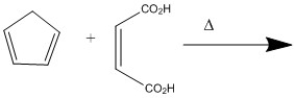
Correct Answer

verified
Correct Answer
verified
Essay
Give the product for the following [1,5] sigmatropic rearrangement. ![Give the product for the following [1,5] sigmatropic rearrangement.](https://d2lvgg3v3hfg70.cloudfront.net/TB1830/11ea7d6e_077a_6e91_8568_79e37f40d98b_TB1830_00_TB1830_00.jpg)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major product of the following reaction? 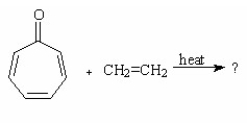
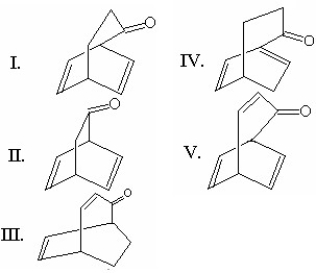
A) I
B) II
C) III
D) IV
E) V
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Under thermal conditions, (2E,4E) -hexadiene undergoes a ________ ring closure to yield ________.
A) conrotatory, cis-3,4-dimethylcyclobutene
B) conrotatory, trans-3,4-dimethylcyclobutene
C) disrotatory, cis-3,4-dimethylcyclobutene
D) disrotatory, trans-3,4-dimethylcyclobutene
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Describe a Claisen rearrangement.
A) [3,3] sigmatropic rearrangement of a 1,5-diene
B) [1,3] sigmatropic rearrangement of an alkene
C) [3,3] sigmatropic rearrangement of an allyl vinyl ether
D) [1,5] sigmatropic rearrangement of a 1,3-diene
E) [2,3] sigmatropic rearrangement of an alkene
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Under photochemical conditions, is an [2+2] cycloaddition suprafacial or antarafacial?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When an allyl cation undergoes a cycloaddition under photochemical conditions with ethylene (bonds forming at the ends of the π-systems) , the cycloaddition is described as ________.
A) [2+2] and suprafacial
B) [2+4] and suprafacial
C) [2+2] and antarafacial
D) [2+4] and antarafacial
E) [2+3] and antarafacial
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is/are the product(s) of the following 1,3-hydrogen shift? 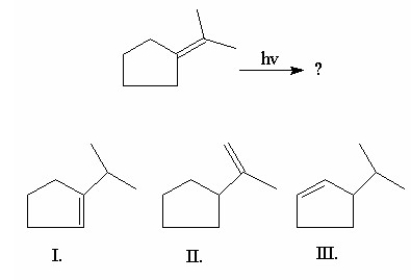
A) I
B) II
C) III
D) I and II
E) I and III
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Essay
What characterizes a pericyclic reaction?
Correct Answer

verified
Pericyclic reactions involve c...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Give the conditions for 1,5-hydrogen shifts.
A) suprafacial rearrangement
B) antarafacial rearrangement
C) thermal conditions
D) photochemical conditions
E) all of the above
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 92
Related Exams